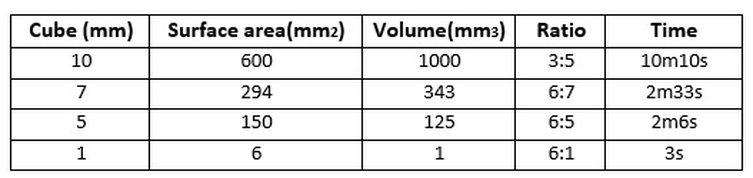
An experiment to investigate the effect of surface area : volume ratio on the diffusion rate.2/9/2016 The objectives of this experiment was to calculate the surface area to volume to ratio of pieces of agar and determine what effect this ratio had on the rate of diffusion. Equipment:
What happens when the agar is placed in the HCl? As time goes on the colour of the agar block becomes increasingly orange as it loses its pink colour. The reason for this is because of the hydrochloric acid diffusing into the block and making it lose its colour. The acid travels from an area of high concentration of HCl (in the test tube) to an area of low concentration of HCl (the agar cube) along a concentration gradient and the point at which the original colour has completely gone is when an even distribution of particles has been achieved. This experiment was designed to then see how this diffusion process occurred in different sized cubes and to investigate the difference I looked at the diffusion rate, i.e. how long it took for the pink colour to disappear. My prediction would be that the rate of diffusion would speed up as the surface area to volume ratio was increased, i.e. the cube was made smaller. Conclusion
From these results I can see that the surface area to volume ratio ad an effect on how fast diffusion occurred. As the agar cubes were cut smaller the ratio of surface area to volume became larger and this affected the diffusion rate by making it faster. The results show a clear trend that supports my knowledge on how the diffusion rate speeds up when there is a larger surface area to volume ratio. The time decreased which highlights how diffusion occurred quicker as there was more surface area in relation to its volume over which the HCl could diffuse over. Another reason for this increase in rate was due to the fact that the HCl particles had to travel longer distances in those agar cubes which had a larger volume in comparison to its surface area. Overall my results support the idea that as the surface area to volume ratio increases so does the rate of diffusion. This experiment relates to real life because single-celled organisms use diffusion as their means of getting the substances they need and expelling those they don’t. Diffusion works efficiently for single-celled organisms such as amoeba due to a large surface area to volume ratio as there is a relatively large surface area over which substances can diffuse into or out of the organism. This is demonstrated in my experiment as the smaller cubes with a larger surface area to volume ratio resemble single-celled organisms. My results also support the idea that large organisms cannot use diffusion as their means of transporting substances in and out, as shown by the 10mm by 10mm cube which took 10 minutes and 10 seconds for the HCl to diffuse all the way through. Diffusion would not be efficient enough for larger organisms as the surface area is not large enough for substances to diffuse over, as a result they require a complex transport system. This experiment does have its limitations as the organism’s cell is represented by a lifeless agar cube. It lacks a semi-permeable membrane and only allows for simple diffusion unlike real-life membranes. Cell surface membranes are much more sophisticated as they consist of a combination of phospholipids, carbohydrates and carrier/channel proteins. As well as this the shape of the agar cube does not resemble an organism’s cell. Apart from these limitations this experiment is a good way of demonstrating the effect of surface area to volume ratio on the rate of diffusion.
4 Comments
John Filter
9/26/2016 09:45:09 am
Do you know what the different variables are? and also what can be done to ensure it is a fair test? thanks
Reply
Alcina
2/1/2021 09:38:32 am
The independent variable is the size of the agar cubes. Dependent is how long for the colour to vanish.
Reply
Leave a Reply. |
Ciara Branagan
Archives
October 2016
|


 RSS Feed
RSS Feed
